A CTO's Journey Into Continuous Biomanufacturing
A conversation with Chris Hwang, chief technology officer, Transcenta Holding Ltd.

At a recent Evaluating BioPharma virtual event, moderator John Bonham-Carter asked Chris Hwang, chief technology officer at Transcenta Holding Ltd., about Chris’ journey and experiences with continuous bioprocessing thus far in his career.
Chris, we first met when you were vice president at Genzyme. It’s a fascinating company; it was the first to publicly state the direction and the thought process about going continuous. There were perfusion processes that needed updating, but you took it further than that. Can you talk about your thought process and why you decided to take those extra steps? What was important to you as you debated that internally?
Early on, back in 2009 or so, we already had this vision of continuous process from looking at how other industries had gone from batch to continuous as they matured, with all the advantages. Our upstream process was already perfusion, we adopted alternating tangential flow (ATF) for cell separation and harvest fluid clarification, and our downstream team was already developing a continuous chromatography system. As a result, it became obvious that we could connect the two together to gain a lot of the benefits in terms of removing the clarification step as well as the large storage tank for clarified harvest fluid, making the process much more streamlined and economical. So that was really the genesis of implementing a continuous integrated capture chromatography system and that was important for us because it simplified the operation quite significantly.
At the time, equipment didn't really exist for the integrated capture, at least not in our industry, so that was quite a risk, as you had to invent a unit operation. How did you think about that risk and the eventual outcome? Did you feel it was justified at the end of it?
We were managing a lot of risks, not just downstream, but upstream too, but on the downstream side we were fortunate to have a very innovative team that was already working on aspects of continuous chromatography. Our downstream team was putting together chromatography columns to try to simulate continuous multi-column systems. They did a lot of lab work to demonstrate it worked, and generated a lot of data to support that this technology can be very robust, certainly focusing on process understanding and equipment/automation understanding. We partnered with GE at the time, which is one of the industry leaders in developing bioprocessing equipment automation, to make these things become implementable in the pilot GMP environment. From that perspective and, just like with any innovation, there was a lot of risk involved.
We take risk management very seriously. We weren’t just generating very good data, but we also did a lot of the FMEA analysis to mitigate all the high risks in implementing something new like this. Then we got the unit in from GE and connected it with our single-use bioreactors and did multiple runs, which demonstrated the system worked as intended. We then generated a lot of very useful data that we used to approach FDA and EMA to get their support and feedback.
A combination of internal and external validation eventually got us to the point where we felt we had the information we needed to move forward. Given the advantages that I already talked about for continuous, it then became obvious that we could convince our management to move forward with this technology.
It’s interesting that you engaged with the regulatory authorities as you went through that process. So, that was one element of mitigating that risk, that you weren't going too far out on a limb, that you understood the data that was going to be required to validate the process. In the end, the facility got built and the experience was very valuable. Is that now deemed a success?
It was highly successful; just imagine that you can replace your four 2,000-liter stainless-steel perfusion bioreactors with one or two 50-liter single-use bioreactors with about two orders of magnitude smaller capture column compared to our original commercial scale, and with the whole integrated system fully automated. This gives you a sense that it's a successful outcome and the success was measured by, firstly, convincing management to move forward, which was a daunting task. However, more important is to get regulatory approval for these types of technologies. There are more coming; my understanding from my colleagues at Sanofi is they’re using this platform for all future products. To me, that's what success looks like – to develop the technology and implement it and, ultimately, benefit patients.
You're now at Transcenta, where you have a whole set of new molecules and processes, and you've taken a series of extra steps on that continuous line. What does fully continuous mean at Transcenta and why have you made those decisions?
The situation in China is very different and I want to explain what drives this. There is significant drug pricing pressure in China, so any drug that comes to China, its price will typically drop at least 50%. Even if domestic products are highly competitive, for you to list your drug in the National Reimbursement List you are required to significantly reduce your drug price. Consequently, low cost of goods (COGs) is not only a competitive advantage for us, in many cases it's a go or no-go decision.
To get the low COGs, that's where process intensification becomes very important. We chose continuous, or a version of the continuous process, and put together a five-year plan to get us there.
We chose a continuous perfusion, steady state process and integrated it with a capture technology, followed by a flow-through polishing system that we're collaborating on with an industry partner. The whole process is not fully end-to-end continuous because our facility has a relatively small footprint and we don't want to tie these new technologies to one product at a time. We want to be able to process multiple reactors and use these technologies to de-bottleneck our facilities. So, we decided to implement a hybrid continuous platform to allow us to maximize the output from the highly flexible and small facility that we’ve built.
So, you immediately had to consider the cost and evaluate each new operation to determine which would be continuous and which wouldn’t. You're not a huge pharmaceutical company with a huge portfolio, so wouldn't you have reached Phase 1 trials faster with batch operations without this investment? Why is continuous best even as you develop the process in preclinical phases?
When I say continuous, I don't mean that we go all in for our early-stage programs. We don't go all in from the very beginning because we're still developing the technology to ensure it's mature and ready to be implemented; we do it incrementally to manage risks. For instance, we may start with fed-batch or perfusion, but our downstream will be batch.
Then we implement as the technology is ready, i.e., we rely on process change during the product development to ensure that the end goal, our PPQ or commercial process, is where we want it to be. For instance, we recently got FDA and CDE approval to switch from fed-batch to perfusion for one of our molecules based on our comprehensive comparability data package. Our philosophy is we don't want to risk our programs, but we want to find ways to take calculated risks, while keeping our end goal in mind.
To focus in on those early stages, fed batch is maybe a two-week process and you move that into perfusion as quickly as you can, but that could double or triple your process development time. How do you avoid extending that time? Is there an extension of time that is worthwhile or is that a misplaced way of stating how you develop a lengthy perfusion process?
A very important criteria for us is that developing this new platform doesn't impact our timeline. In fact, we developed the perfusion as a plug and play platform and typically we run our Phase 1 process at no more than 21 days. However, because it's plug and play, there's no development when we go to Phase 1 – you plug in your cell line and run it for the duration that you desire. We can typically achieve, I would say, from top clones to the process block in less than 10 weeks, which is very, very fast.
Typically, our timeline from DNA to CMC package for IND filing is still around 10 months. It hasn't slowed us down; in fact, I think it might accelerate it a little bit in terms of our time to clinic when compared to fed-batch. From that standpoint, we don't think process development would extend the timeline. There's certainly more work to be done later in product development, but that's not going to become the rate limiting element, as that tends to occur more around the clinical stages. We don't see how implementing continuous technology is going to negatively impact our speed.
It's always good to have a vision, but executing on the vision is the hard part. How are you delivering against your five-year plan at Transcenta?
We started this five-year plan in 2018 when our facilities opened in China and we’re getting close to being done; 2023 would be five years. COVID has delayed the overall time by about six months, but we are well into it.
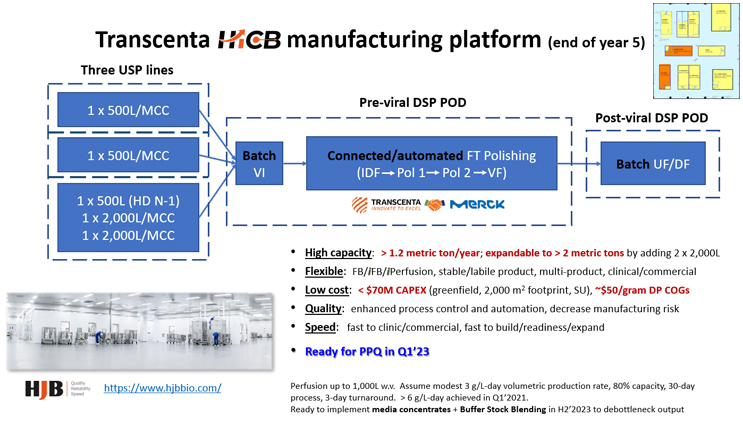
We started on this five-year plan by building the foundation for the perfusion. We had to build everything from scratch, from the cell line to media and the perfusion platform, but within about year and a half we had completed development of this platform and we are now applying it to every single one of our internal products.
We also partnered with MilliporeSigma to develop our fully automated single-use flow-through polishing system that combines four unit operations into one, which significantly decreases the cycle time of downstream processing, including the integrated multi-column system (MCC) that we’ll implement as well.
So, this platform is almost ready. The combo system is currently in-house since late 2021, and, unfortunately, I can’t share a lot of information, but I will in the future. This system will be fully ready for GMP operations very soon this year and then the MCC will be later in the year. Our goal is to have intensified DSP ready to go for the PPQ runs in Q1 2023.
So, you've got some smaller bioreactors and some larger bioreactors feeding into the same pods downstream. So that's four times the volume coming through; how do you match that up?
As we decided not to do end-to-end, the hold point, in this case, would be Protein A. From our experience, Protein A is fairly stable and the volume is manageable. This allows us, for any product going through, whether it is fed-batch, intensive fed-batch, or in this case, continuous perfusion, to quickly run through pre-viral downstream processing in a very short amount of time. Then you follow it with the post-viral processing.
The other reason we didn't go fully continuous is that we don't want to tie up our pre-viral and post-viral downstream PODs to just one product at a time for the duration of perfusion run. We want to maximize output from our downstream clean rooms by increasing the speed of processing and support multiple products. From typically four or five days of operation, we can make it in one or two days. That allows us to handle everything that's coming from our upstream processes, regardless of products and type of processes.
There are two technical points I want to finish on. With your development system being the same size as the manufacturing scale system, it allows you high confidence in that tech transfer and eliminates risk and time as you do that. Is that your experience, too? Is that how you run it in the development scale?
It depends on how you intensify your processes. From our standpoint, our lab scales are typically benchtop lab scales, so they're typically 2 liters or so, but when we go to pilot, or in this case Phase 1 manufacturing, commercial, the scale will be much larger.
I would say going from Phase 1 process to commercial, there's little to no scale-up, which significantly reduces manufacturing risks.
In what I would call quite a sophisticated large perfusion facility, you've got a smaller number of products compared to large pharma. How do you balance that capacity?
First, our facility is not large. The manufacturing ballroom is only about 2,000 square meters and our facility was not built at a full capacity up front. Our capacity can expand over time, so you don't have to build the full capacity at the outset. With increasing demand, whether it is from our internal pipeline or supporting our CDMO clients, we will increase our capacity very quickly as needed to meet the projected demand.
So that's why these types of facilities are going to become very important. You need to be flexible, not just to support different type of molecules, but also so you can build and expand very quickly to mitigate the capacity risk.
